Scientific discovery is often imagined as a solitary eureka moment—a singular mind or laboratory unlocking nature’s secrets in isolation. But history tells a different story. Some of the most transformative breakthroughs have happened twice, independently, and almost simultaneously.
In the 17th century, both Isaac Newton and Gottfried Leibniz developed the foundations of calculus, entirely unaware of each other’s work. Two centuries later, Charles Darwin and Alfred Wallace, working oceans apart, each arrived at the same theory of natural selection, prompting Darwin to publish On the Origin of Species.
These moments of “multiple discovery” remind us that science is a collective enterprise. Progress is guided by shared questions and evolving tools, eventually creating a historical moment in which certain ideas become possible and parallel paths converge on the truth. Medicine, too, has borne witness to this phenomenon.
By the early 1950s, physiology was undergoing a quiet revolution. Biochemistry, once peripheral, had become key to answering fundamental questions about how the body worked. With the invention of new tools, such as electron microscopes, phase-contrast imaging, and refined staining techniques, longstanding mysteries were finally within reach.
On May 22, 1954, Nature published what may be one of the most serendipitous dual discoveries in the history of medicine. In a remarkable coincidence, two unrelated researchers with the same last name—Andrew Huxley and Hugh Huxley—each submitted independent studies from different institutions describing what would soon become canon: the sliding filament theory of muscle contraction. Their findings reshaped our understanding of how skeletal and cardiac muscle generate force and remain foundational to the understanding of physiology today.
In the first paper, Interference Microscopy of Living Muscle Fibres, Andrew Huxley, already renowned for the Hodgkin-Huxley model of action potentials, and his colleague Rolf Niedergerke used interference microscopy to visualize live, unfixed frog muscle fibers in real time. They observed that during contraction, the I-band shortened while the A-band remained unchanged. This critical finding suggested a dynamic interaction between interlaced structural components, rather than a uniform shortening of the whole muscle fiber.
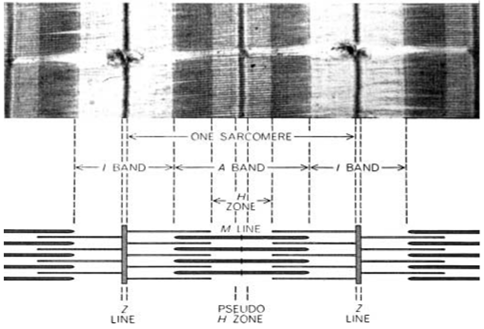
The second paper, Changes in the Cross-Striations of Muscle during Contraction and Stretch and their Structural Interpretation, was authored by Hugh Huxley and Jean Hanson. Their approach differed, using phase-contrast and electron microscopy on fixed rabbit muscle. Even so, they observed comparable structural changes but interpreted them through a different lens. They, too, proposed that actin and myosin filaments slide past one another to generate contraction (Figure 1), overturning older “folding” theories.
Figure 1 : H. Huxley’s electron micrograph (top) and schematic depiction of the sarcomere (bottom) from 1954. Myosin is found in the thick filaments and actin in the thin filaments.
Though their methodologies differed—frog versus rabbit muscle, unfixed versus fixed tissue, interference versus electron microscopy—their conclusions aligned remarkably. Both teams proposed a model in which two interlaced sets of structural components, thin (actin) and thick (myosin) filaments, slide past each other to shorten the sarcomere, the fundamental unit of muscle contraction. The terminology they introduced, including A-band, I-band, H-zone, and Z-line, is still widely used in physiology texts today.
Hugh Huxley and Hanson went a step further. They proposed that myosin heads form transient linkages with actin, powered by repeated cycles of ATP hydrolysis, generating the mechanical force that drives contraction. This hypothesis became the foundation of the cross-bridge cycle. Hugh Huxley refined and formally published the theory in the 1960s, completing the modern understanding of how skeletal and cardiac muscles generate force.
The fact that two unrelated scientists named Huxley, working independently at different institutions, using different microscopes to study different species with distinct techniques, published near-identical conclusions in the same issue of Nature is more than a historical footnote. It’s a testament to how scientific discovery is often concurrent—driven by technological advances, shifting perspectives, and the convergence of opportunity and inquiry.
In the age of cardiac MRI, single-cell RNA sequencing, and CRISPR-edited models of heart disease, the tools we use and questions we ask have grown exponentially more complex—reflected in the rapid expansion of fields like molecular biology and biochemistry. History reminds us that even seemingly small questions, once answered, eventually create an environment that permits new ideas to emerge, often simultaneously and independently, as if by coincidence. The dual discovery of the sliding filament model by the two Huxleys serves as a memorable example. Scientific progress rarely occurs in isolation, but instead through the remarkable convergence of independent discoveries—proof that science advances as a shared, ever-evolving journey toward the truth.
Picture citation:
Huxley HE. Fifty years of muscle and the sliding filament hypothesis. European Journal of Biochemistry. 2004;271(8):1403-1415. doi:10.1111/j.1432-1033.2004.04044.x

Austin Mineer
Austin Mineer is a fourth year medical student at the University of Arizona College of Medicine-Phoenix. He graduated from University of Utah, majoring in Biochemistry and minoring in Mathematics. He is applying to Internal Medicine with plans to further pursue Cardiology. Austin is an avid music collector with over 600 albums on CD and vinyl and enjoys going to concerts, hiking, learning Japanese, and playing Smash Bros in his free time. He has addictions to matcha and Mexican food. Please feel free to reach out at austinmineer@arizona.edu.